NOTCH Signaling Antibodies
The NOTCH pathway is a fundamental signaling system allowing neighboring cells to communicate and perform their proper developmental role. NOTCH proteins mediate critically important cellular functions through direct cell-cell contact. NOTCH signaling involves gene regulation mechanisms that control multiple cell differentiation processes during embryonic and adult life.
NOTCH signaling also has a role in:
- Neuronal function and development
- Stabilization of arterial endothelial fate and angiogenesis
- Regulation of crucial cell communication during formation of the valve primordial and ventricular development and differentiation
- Cardiac valve homeostasis
- Timely cell lineage specification of both endocrine and exocrine pancreas
- Influencing of binary fate decisions of cells
- Expansion of the hematopoietic stem cell compartment during bone development
T cell lineage commitment from common lymphoid precursor
Faulty NOTCH signaling is implicated in many diseases including T-ALL (T-cell acute lymphoblastic leukemia), CADASIL (Cerebral Autosomal Dominant Arteriopathy with Sub-cortical Infarcts and Leukoencephalopathy), MS (Multiple Sclerosis), Tetralogy of Fallot, Alagille syndrome, and many cancers.
Rockland products can help in a variety of Notch signaling research areas. Notch receptors are single-pass transmembrane proteins composed of functional extracellular (NECD), transmembrane (TM), and intracellular domains. In the signal-receiving cell, γ-secretase (also involved in Alzheimer’s disease) releases the NICD from the TM (S3 cleaved notch), which translocates to the nucleus where it associates with the CSL (CBF1/Su(H)/Lag-1) family transcription factor complex, resulting in subsequent activation of the canonical csl notch target genes Myc, p21 and HES-family members.
NOTCH Signaling Pathway
Notch signaling pathways are a booming area of pharmacological research, due largely to the direct connection to human disease intervention. Notch receptor activating mutations leading to nuclear accumulation of NICD is common in adult T-cell acute lymphoblastic leukemia and lymphoma. In addition, loss-of-function Notch receptor and ligand mutations are implicated in several disorders, including Alagille syndrome and CADASIL, an autosomal dominant form of cerebral arteriopathy. Research utilizing antibodies and proteins found within the notch signaling pathway holds great promise in cancer treatment as well, making these products highly valued in the scientific community.
The evolutionary highly conserved notch signaling pathway regulates cell-fate determination during development and maintains adult tissue homeostasis. Notch signaling is mediated by proteolysis. Regulation of the amount and timing of the notch signal is managed by posttranslational modifications to ligands and receptors and their trafficking.
The Notch receptors are single-pass type I transmembrane heterodimer proteins. They encompass a functional extracellular truncation (NEXT), transmembrane (TM), and intracellular (NICD) domain. The Notch receptor is activated by binding to a ligand presented by a neighboring cell. Maturation of Notch happens in the ER and Golgi. Protein fucosylation is essential for the functional receptor and occurs via the chaperone O-fut in the ER. Different patterns of fucose on Notch change the aptitude of specific ligands to activate Notch. Fucose extension is performed by Fringe. Upon proteolytic cleavage by Furin at site 1 (S1) Notch is transported to the cell surface.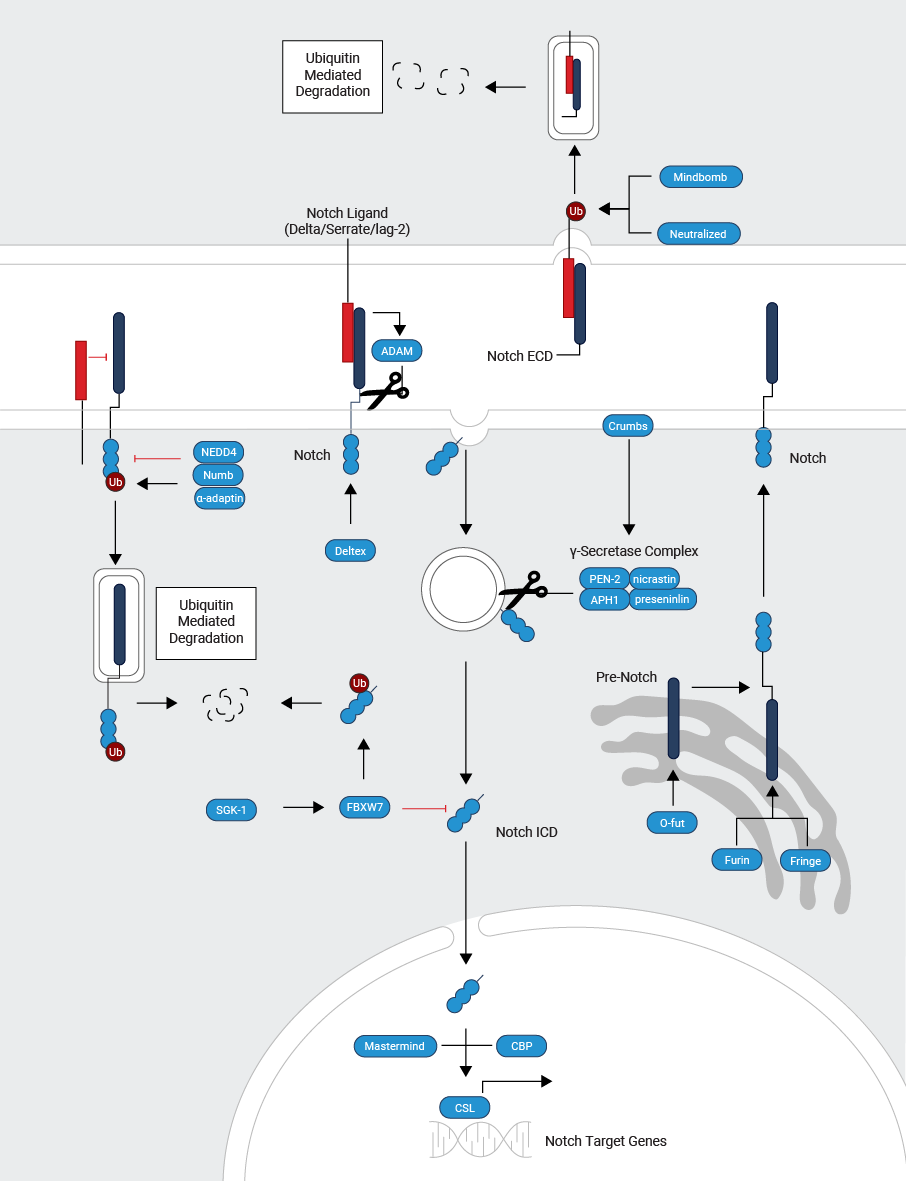
Delta and Jagged are the major classes of Notch ligands. They are type I transmembrane proteins and are ubiquitinated by Neur and Mib (E3 ubiquitin ligases), triggering Epsin mediated endocytosis. Modification of the Notch ligands induces recycling of the ligand to the cell surface in a Rab11 dependent process. Fruitful receptor-ligand interactions depend on the glycosylation state of Notch. Ligand endocytosis is thought to produce partial or complete domain dissociation, thereby exposing Notch to cleavage at site S2 by ADAM metalloproteases. Upon cleavage, the Notch extracellular domain is trans-endocytosed into the signal-sending cell (neighboring cell). The membrane-anchored NEXT fragment is recognized by Nicastrin (NCT), which transfers NEXT to the active site of γ-secretase. γ-secretase is an enzymatic complex composed of presenilin (PS), NCT, PEN2, and APH1. γ-secretase cleaves the Notch transmembrane domain sequentially starting near the cytosolic surface (sites S3 and S4) to release the Notch intracellular domain (NICD) and Nβ peptides, respectively.
NICD enters the nucleus to facilitate transcription. NICD binding to the DNA-binding protein CSL triggers an allosteric change that facilitates the displacement of transcriptional repressors. The NICD/CSL interface is recognized by Mastermind (MAM), and this complex recruits Co-activators (Co-A) like histone acetylases (HATs), to assemble an active transcription complex on target promoters. When NICD is not present, CSL associates with ubiquitous Co-repressor proteins (Co-R) and histone deacetylases (HDACs) to block transcription of target genes.
NOTCH Pathway Antibodies
| Product | Clonality | Reactivity | Applications |
| APH1 Antibody | Polyclonal | Human, Mouse | WB, ELISA |
| APH1 Antibody | Polyclonal | Human, Mouse, Rat | WB, IHC, ELISA |
| CREBBP Antibody | Polyclonal | Human, Mouse | WB, ELISA |
| HDAC1 (near C-terminus) Antibody | Polyclonal | Human, Mouse, Primate, Rat | WB |
| HDAC1 (C-terminus) Antibody | Polyclonal | Human | WB, IF, IHC, ELISA |
| HEY1 Antibody | Polyclonal | Human, Mouse | WB, IHC, ELISA |
| Jagged 1 Antibody | Polyclonal | Human, Mouse | WB, IF, IHC, ELISA |
| PEN2 Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, ELISA |
| PEN2 Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| Presenilin1 Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| Nicastrin Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| Nicastrin Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| NOTCH 1 Antibody | Polyclonal | Human, Mouse | WB, IF, IHC, ELISA, IP, Dot Blot |
| NOTCH 1 Antibody | Polyclonal | Human | IHC, ELISA |
| NOTCH 2 Antibody | Polyclonal | Human | WB, IHC, ELISA |
| NOTCH 2 Antibody | Polyclonal | Human | WB, IF, IHC, ELISA |
| NUMB Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| Presenilin1 Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| SkiP Antibody | Polyclonal | Human, Mouse, Rat | WB, IF, IHC, ELISA |
| TACE Antibody | Polyclonal | Human, Rat | WB, IF, IHC, ELISA |