HCP Antibodies
HCP Antibodies
Leverage Rockland’s 15+ years of success generating custom polyclonal host cell protein antibodies (HCP antibodies) to keep your biopharmaceutical development program meeting deadlines and moving efficiently.
Our comprehensive, fully customizable services cover all aspects of anti-HCP antibody generation, from antigen preparation and assay development to documentation and manufacturing, with an end-goal of creating a broad immune response as measured by a high percent of coverage on a 2D Western blot.
Whether you need support from start-to-finish or are only outsourcing small parts of the project, you can be confident when you partner with Rockland.
Choose Rockland when you need quality and efficiency
Rockland’s HCP Antibody service combines expertise with optimized processes to maximize quality, coverage, and speed.
- Consistent—Minimize lot-to-lot variability and ensure long term supply chain with well-managed antisera pools
- High-quality—Maximize repeatability and safeguard quality with our rigorous testing and multi-validation studies
- Scalable—Experience stable large-scale manufacturing with our strong internal processes
- Transparent—Maintain visibility into your project with all development and manufacturing conducted at our US-based facility
- Experienced—From initial intake to manufacturing and shipping, our experienced scientists, project managers, and logistics team ensure accuracy and efficiency
| HCP Antibody Generation Timeline (Months) | ||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Setup | ||||||||
| Immunization & Boost | ||||||||
| Reagent Generation | ||||||||
| Sampling & Analysis | ||||||||
| Reagent Qualification | ||||||||
Fully customize your anti-HCP antibody generation service
Choose all-inclusive anti-HCP antibody generation or select only the services you need.
Antigen Preparation
- Product Platform: E. Coli, Yeast Insect, Plant, & Mammalian
- Fractionation
- Ultrafiltration
- Crosslinking & Conjugation
Antisera Generation
- Standard Protocol
- Size Fractionated Immunogen
- Cascade Immunogen
- Rabbit, Goat, Chicken, Sheep
Reagent Characterization & Analysis
- ELISA
- 1D SDS & Western Blot
- 2D SDS & Western Blot
- Reagent Qualification
- Coverage Analysis & Characterization
Assay Development
- Scouting Study
- Bridging Study
- Process Specific HCP Assays
- Multiple Platform Support
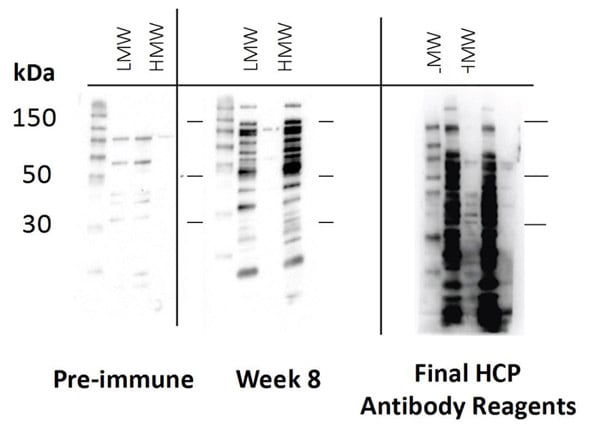
See the strong, uniform signal of a final HCP antibody reagent
We use 1D SDS-PAGE followed by Western blot analysis on test and production bleeds to evaluate the host antibody-mediated immune response to HCP lysate.
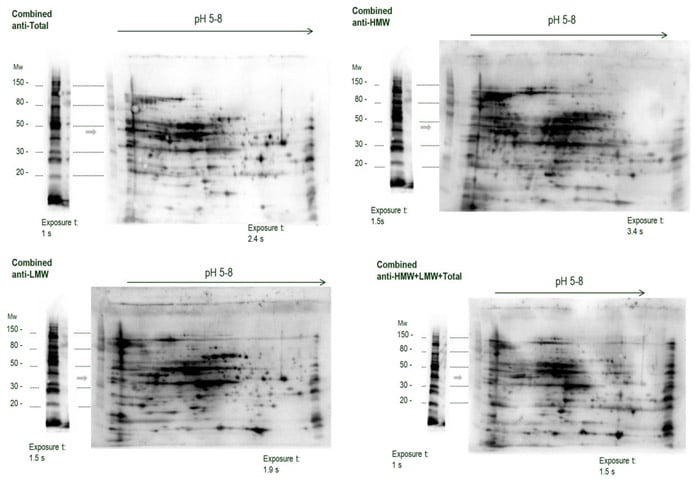
We also evaluate HCP antibodies with 2D PAGE and Western blotting
We can also conduct 2D SDS-PAGE followed by Western blotting. 2D separation can deliver more complete insight into an anti-HCP antibody’s coverage of proteins in the host cell lysate than 1D SDS-PAGE and Western blotting
Learn more
Impurity Detection ELISA Kits
Mitigate risk of host cell protein impurities with generic HCP ELISA kits developed and validated in partnership with Cytiva Life Sciences.
Learn moreReady for HCP antibody production?
We have a wealth of experience ready to achieve your program’s needs.