Oligonucleotide Analysis using ModDetect®
Oligonucleotide Analysis using ModDetect®
Rockland’s immunoassay development team partners with you to design and execute in vitro immunoassays that support the analysis of oligonucleotide therapeutics across discovery and preclinical development. Using ModDetect® antibody panels, we generate high-quality data on biodistribution, intracellular localization, quantification, and trafficking, tailored to your drug modality and analytical objectives to enable faster, more confident candidate triage.
Bioanalytical Challenges in Oligonucleotide Drug Development
Assessing oligonucleotide therapeutics during discovery and preclinical development presents unique analytical challenges. Advancing the strongest candidates requires robust assessment of absorption, distribution, metabolism, and excretion (ADME) alongside a clear understanding of pharmacokinetic (PK), pharmacodynamic (PD), and immunogenicity profiles.
Many conventional analytical methods are limited by sequence dependence, complex probe design, or reduced sensitivity for chemically modified oligonucleotides. These constraints can slow data generation and complicate comparisons across candidates and modalities.
Immunoassay-based approaches offer a powerful orthogonal alternative, enabling sensitive, modification-specific detection that supports decision-making throughout the preclinical pipeline.
Exploring Stereoisomer Analysis?
ModDetect®-enabled Immunoassay Services
Rockland’s immunoassay development team designs and executes custom ModDetect-enabled assays tailored to your oligonucleotide therapeutic and analytical objectives. Using our proprietary antibody panels that recognize key chemical modifications, such as phosphorothioate (PS), 2′-O-methoxyethyl (2′-MOE), and 2′-O-methyl (2′-OMe), we deliver sequence-independent data across relevant biological matrices.
Our services support:
- Biodistribution and tissue localization
- Intracellular localization and trafficking
- Quantitative detection in cells, tissues, and biofluids
- Early assay feasibility and reagent performance evaluation
By outsourcing assay development and execution to Rockland, teams gain access to validated workflows, expert interpretation, and actionable recommendations that accelerate candidate selection and reduce analytical bottlenecks.




Bioanalytical Data Supporting Oligonucleotide Development
Representative examples of bioanalytical data generated using ModDetect™ are shown below. These data illustrate how ModDetect antibodies are applied across multiple assay formats to support the evaluation of oligonucleotide therapeutics, including assessments of biodistribution, intracellular localization, quantitative detection, and binding characteristics. Assay design and readouts are customized based on oligonucleotide chemistry, analytical objectives, and sample type.
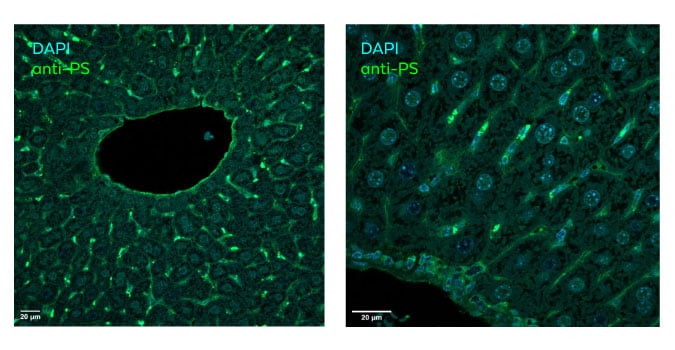
In vivo Biodistribution in Tissue
ModDetect® antibodies enable visualization of oligonucleotide biodistribution in tissue sections, supporting assessment of delivery and tissue exposure in preclinical models.
Explore the Poster: Characterising antibodies targeting antisense oligonucleotide (ASO) modifications for quantification of intracellular trafficking and in vivo distribution
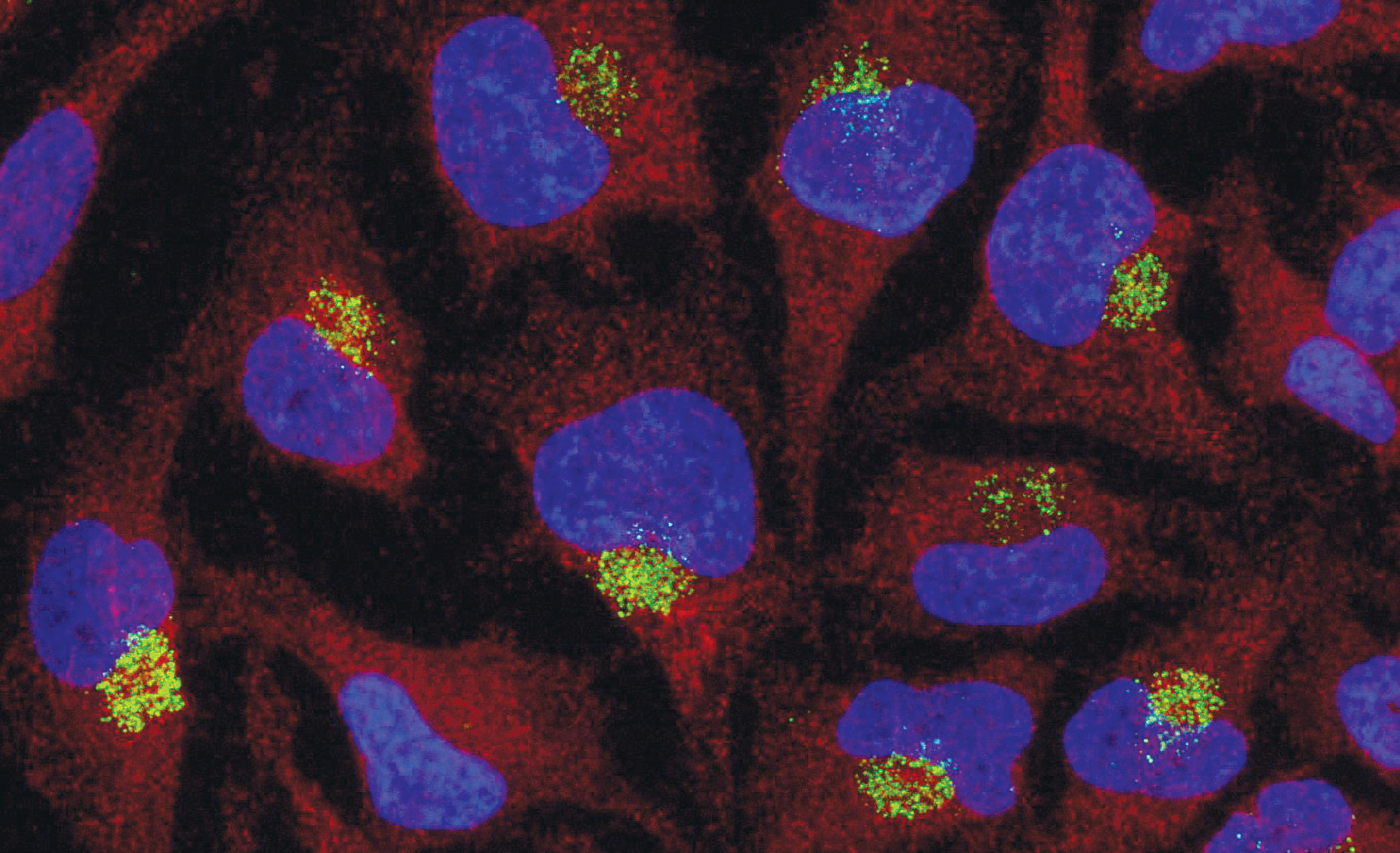
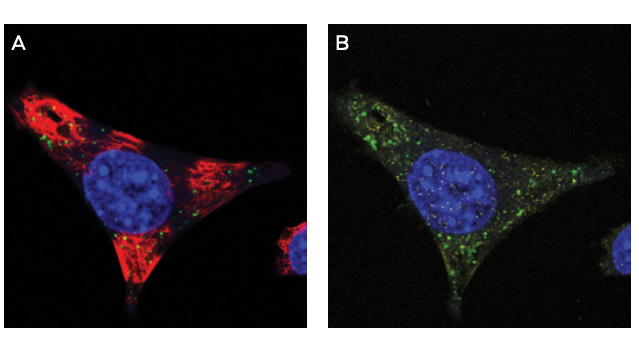
Intracellular Localization in Cells
Representative immunofluorescence image showing ModDetect®-based detection of intracellular oligonucleotide localization, supporting uptake and trafficking assessments in cell models.
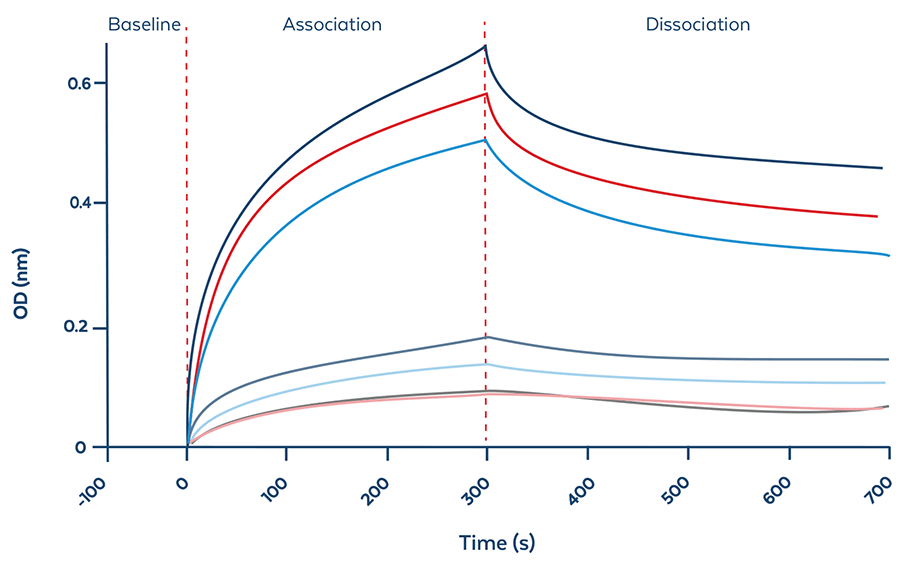
BLI Evaluation
Bio-layer interferometry (BLI) analysis enables evaluation of ModDetect® antibody binding characteristics to oligonucleotide therapeutics, supporting reagent selection and assay development.
| Parameter | Result |
| Affinity Constant (KD) | 1.25 nM |
| Association Rate (Ka) | 6.25 x 105 M-1s-1 |
| Dissociation Rate (Kd) | 7.81 x 10-4 s-1 |
ModDetect® Analytical Capabilities
Rockland offers ModDetect-enabled immunoassays tailored to specific analytical objectives, sample types, and oligonucleotide modalities. The table below highlights representative assay formats and applications supported by our team.
| Analytical Objective | Assay Format | Sample Types | Oligo Modalities |
| Biodistribution | IHC | Mouse tissue | ASO, siRNA, aptamer, mRNA |
| Intracellular Localization | IF | Cell culture, mouse tissue | ASO, siRNA, apatamer, mRNA |
| Penetration Studies | IF 3D | Spheroids, organoids, 3D cell culture, tumor tissue | ASO, siRNA |
| Compartmentalization Studies | IF | Cell culture, tissue | ASO, siRNA |
| Passenger vs. Guide Strand Differentiation | IF | Serum or plasma | siRNA |
| Anti-Drug Antibody Binding | ELISA | Serum or plasma, cell culture supernatants | ASO, siRNA |
| Immunogenicity Studies | ELISA | Serum or plasma, cell lysates | ASO, siRNA, aptamer, mRNA |
| Quantification | ELISA | Tissue lysates, biofluids | ASO, siRNA, aptamer, mRNA |
| Binding Protein Identification | IP | Serum or tissue lysates | ASO |
| Potency | In vitro tissue culture | Tissue culture | ASO, siRNA |
| Identification/Separation of Target Cells | Cell sorting | Cell culture | ASO |
Additional assay formats, sample types, and oligonucleotide modalities may be supported. Contact us to discuss your specific analytical requirements.
Let Rockland do the heavy lifting:
Reduce internal workload when analytical resources or expertise are limited
Save development time and focus internal teams on oligonucleotide design and optimization
Gain comprehensive data and expert interpretation from immunoassay specialists
Accelerate candidate triage with reliable, modification-specific insights
Explore Supporting ModDetect® Resources
For teams seeking additional technical context, Rockland provides access to peer-reviewed publications, application notes, and technical posters demonstrating the use of ModDetect® across oligonucleotide modalities and assay formats.