Datasheet is currently unavailable. Try again or CONTACT US
Recombinant Anti-CTLA4 Fab Antibody
400-001-MT1
50 µg
Liquid (sterile filtered)
WB, ELISA, FC
Human
HEK293
Shipping info:
$65.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Product Details
Humanized Recombinant Anti-Human CTLA4 Fab fragment Antibody - 400-001-MT1
Cytotoxic T-Lymphocyte Associated Protein 4, Ipilimumab, Yervoy, recombinant Monoclonal CTLA4, RabMAb CTLA4, CTLA-4, CD152, Insulin-Dependent Diabetes Mellitus 12, Cytotoxic T-Lymphocyte Protein 4, Celiac Disease 3, GSE, CD, Ligand And Transmembrane Spliced, Cytotoxic T Lymphocyte Associated Antigen 4, Cytotoxic T Lymphocyte Associated Antigen 4 Short Spliced Form, Cytotoxic T-Lymphocyte-Associated Serine Esterase-4, Cytotoxic T-Lymphocyte-Associated Antigen 4, CD152 Isoform, CD152 Antigen, CELIAC3, IDDM12, ALPS5, GRD4
Recombinant Monoclonal
IgG Fab
HEK293
Target Details
CTLA4 - View All CTLA4 Products
Human
Recombinant Protein
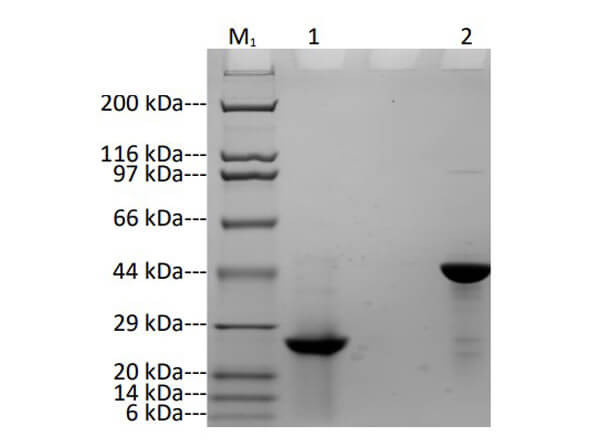
Humanized Recombinant CLTA4 Fab fragment was expressed in HEK293 cells. Purified using one-step purification by HisTrap FF Crude. The purity was estimated to be >95% by SDS-PAGE analysis.
Application Details
ELISA, FC, WB
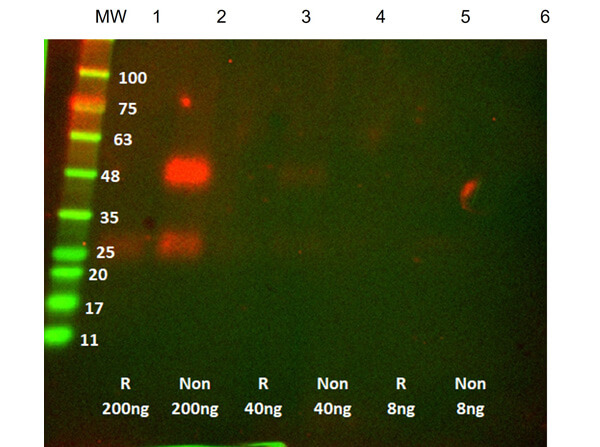
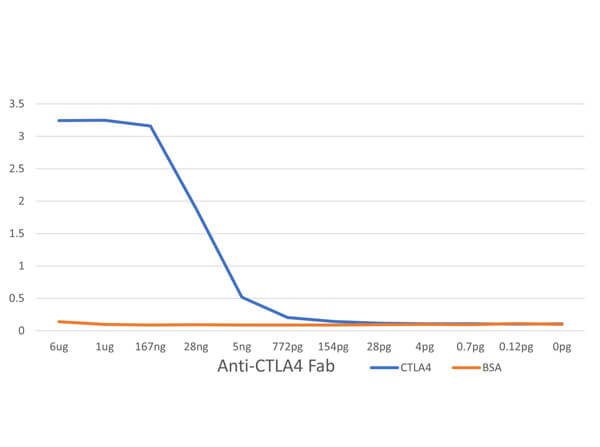
Humanized Recombinant Anti-CTLA-4 Fab fragment Antibody is tested for use in Flow Cytometry, Western Blot, and ELISA. This antibody recognizes structured CTLA-4 and will work in western blot when the protein has not been denatured with DTT or bMe. Although not tested, this antibody could be useful in in IHC and in in-vivo and other cellular assays. Specific conditions for reactivity should be optimized by the end user.
Formulation
0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2
None
30% Glycerol
Shipping & Handling
Wet Ice
Store vial at -20° C. Dilute only prior to immediate use.
Expiration date is six (6) months from date of receipt.
CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4) is also known as CD152 (Cluster of differentiation 152), is a protein receptor that functions as an immune checkpoint and downregulates immune responses. CTLA4 is constitutively expressed in regulatory T cells but only upregulated in conventional T cells after activation – a phenomenon which is particularly notable in cancers. It acts as an "off" switch when bound to CD80 or CD86 on the surface of antigen-presenting cells. CTLA4 is a member of the immunoglobulin superfamily. The protein contains an extracellular V domain, a transmembrane domain, and a cytoplasmic tail. Alternate splice variants, encoding different isoforms. CTLA4 is similar to the T-cell co-stimulatory protein, CD28, and both molecules bind to CD80 and CD86, also called B7-1 and B7-2 respectively, on antigen-presenting cells. CTLA4 transmits an inhibitory signal to T cells, whereas CD28 transmits a stimulatory signal. Intracellular CTLA4 is also found in regulatory T cells and may be important to their function. T cell activation through the T cell receptor and CD28 leads to increased expression of CTLA-4, an inhibitory receptor for B7 molecules. Fusion proteins of CTLA4 and antibodies (CTLA4-Ig) have been used in clinical trials for rheumatoid arthritis. This antibody is made from a humanized Fab fragment of anti-CTLA4, ipilimumab or Yervoy, the first FDA approved immune checkpoint blockade therapy for cancer treatment. Humanized Recombinant Anti-CTLA4 fragment Antibody is useful for researchers interested in cancer research, immunodeficiency, and autoimmune diseases.
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.