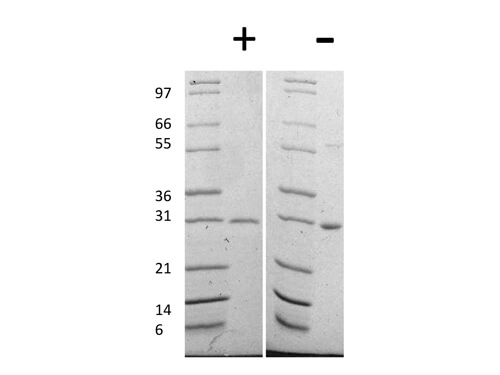
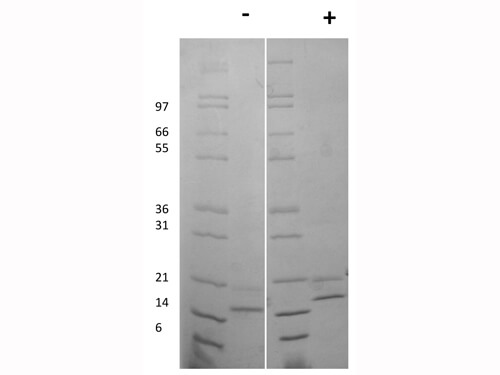
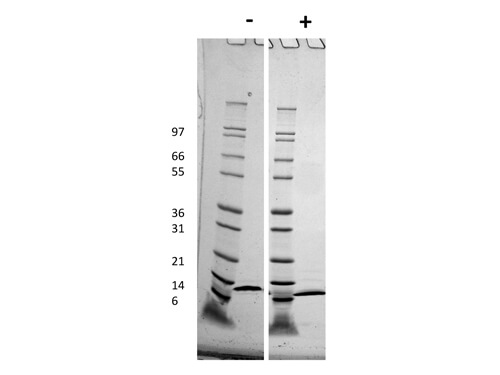
Human IgG Fc
6 References
009-0103
1 mg
Liquid (sterile filtered)
WB, ELISA, IF, Dot Blot, SDS-PAGE, Cellular Assay
Human
Shipping info:
$65.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Product Details
Human IgG Fc Fragment - 009-0103
Human Immunoglobulin G F(c) Fragment, IgG Fc
Human
IgG Fc
Native Protein
Target Details
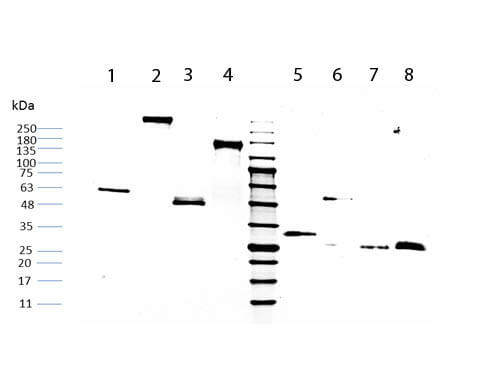
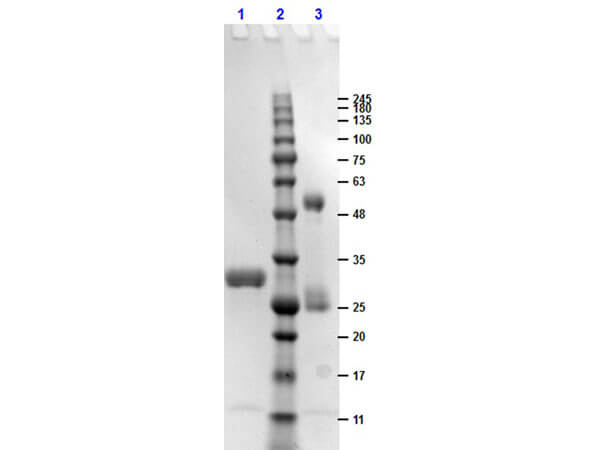
HUMAN IgG F(c) was prepared from normal serum by a multi-step process which includes delipidation, salt fractionation, ion exchange chromatography and papain digestion followed by chromatographic separation and extensive dialysis against the buffer stated above. Assay by immunoelectrophoresis resulted in a single precipitin arc against anti-Human Serum, anti-Human IgG and anti-Human IgG F(c). No reaction was observed against anti-Human IgG F(ab’)2 or anti-Papain.
Application Details
Dot Blot, SDS-PAGE
Cellular Assay, ELISA, IF, WB
- View References
HUMAN IgG F(c) Fragment has been tested by SDS-Page and dot blot and can be utilized as a control or standard reagent in SDS, Western Blotting, and ELISA experiments.
Formulation
1.029 mg/mL by UV absorbance at 280 nm
0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2
0.01% (w/v) Sodium Azide
None
Shipping & Handling
Wet Ice
Store vial at 4° C prior to opening. This product is stable 4° C as an undiluted liquid. Dilute only prior to immediate use. For extended storage mix with an equal volume of glycerol, aliquot contents and freeze at -20° C or below. Avoid cycles of freezing and thawing.
Expiration date is one (1) year from date of receipt.
Human IgG Fc purified protein is a proteolytic fragment of immunoglobulin G (IgG) obtained by limited digestion with the enzyme papain under controlled conditions of temperature, time and pH. Receptors bind the Fc portion of Human IgG and often this fragment is removed from immunoglobulins to minimize receptor binding and lower background reactivity.
Mathew S et al. (2025). Altered Expression of NK Receptors in Racially/Ethnically Diverse and Risk-of-Relapse Pediatric Acute Lymphoblastic Leukemia Patients. Biomedicines.
Applications
FC, FACS, FLOW
Eid L et al. (2025). Both GEF domains of the autism and developmental epileptic encephalopathy-associated Trio protein are required for proper tangential migration of GABAergic interneurons. Mol Psychiatry.
Applications
IF, Confocal Microscopy; Control
Kundu S et al. (2024). Inhibition of the extracellular matrix protein fibulin-3 reduces immunosuppressive signaling in tumor stem cells and increases macrophage activation against glioblastoma. bioRxiv [preprint]
Applications
Blocking
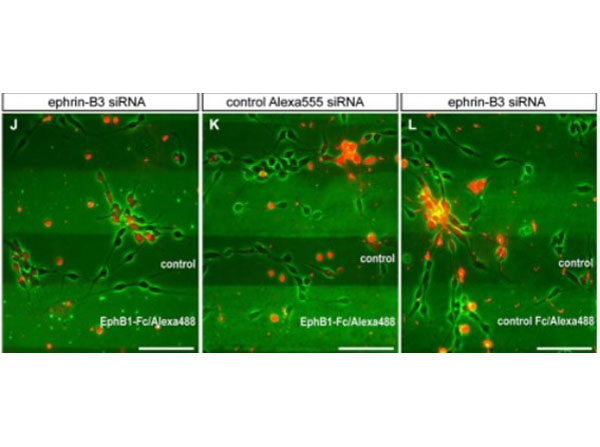
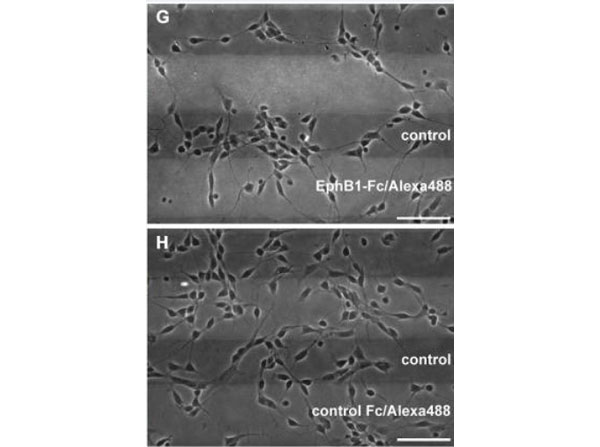
Rudolph, J et al. (2014). A dual role of EphB1/ephrin-B3 reverse signaling on migrating striatal and cortical neurons originating in the preoptic area: should I stay or go away? Frontiers in Cellular Neuroscience
Applications
Other; Cellular assay
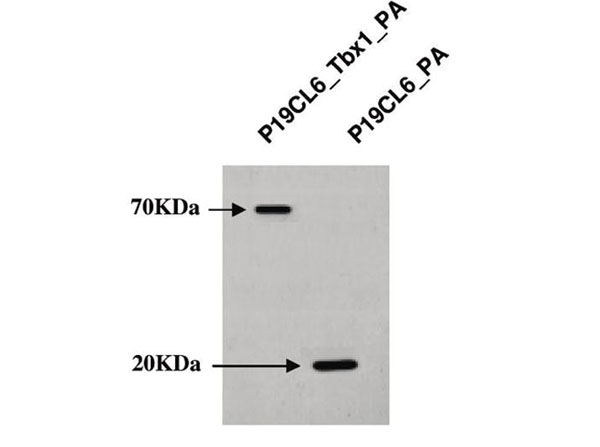
Caterino, M et al. (2009). Transcription factor TBX1 overexpression induces downregulation of proteins involved in retinoic acid metabolism: a comparative proteomic analysis. Journal of Proteome Research
Applications
WB, IB, PCA
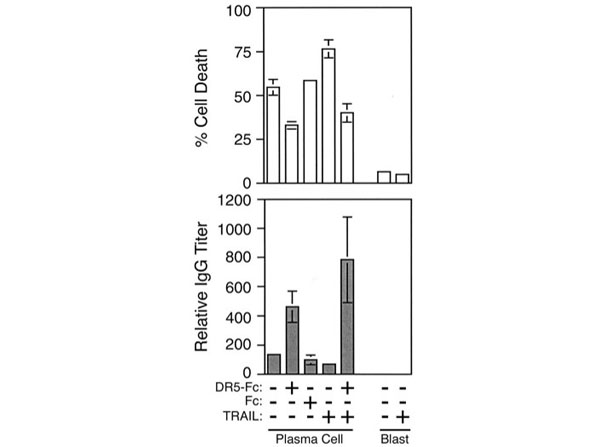
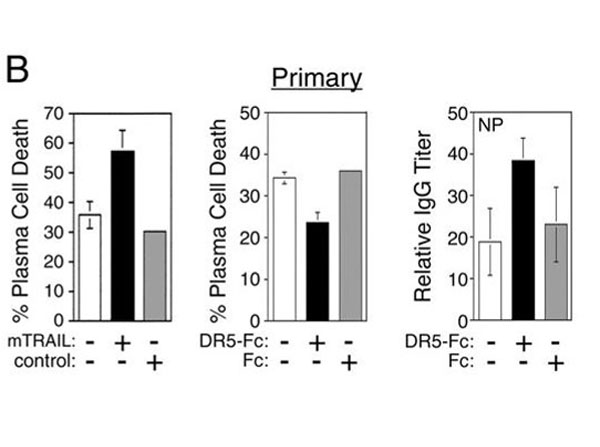
Ursini-Siegel J et al. (2002). TRAIL/Apo-2 ligand induces primary plasma cell apoptosis. J Immunol.
Applications
E, EIA
No test method can provide total assurance that the hepatitis B virus, hepatitis C virus, human immunodeficiency virus, or any other infectious agents are absent. Thus, all blood products, including purified proteins derived from human blood sources, should be handled at Biosafety Level 2 as recommended by the CDC/NIH manual entitled Biosafety in Microbiological and Biomedical Laboratories for potentially infectious human serum, blood specimens or proteins derived from same. Source material for the human blood product supplied to your facility has been tested for the detection of HIV antibody, Hepatitis B surface antigen, antibody to Hepatitis C, HIV 1 antigen(s), antibody to HTLV - I/II, and syphilis by FDA guidelines. All units were found to be non-reactive/negative for these tests. All human blood source material is collected in FDA licensed centers and is tested with FDA approved test kits.; This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.