Flow Cytometry Technique
Flow cytometry (FC) is a technology that allows rapid and quantitative multi-parameter assays on single living (or dead) cells by measuring visible and fluorescent light emission signals. Furthermore, by analyzing the expression of intracellular and cell surface molecules using fluorophore-labeled antibodies, FC provides quantitative information of antigenic, biochemical, and biophysical characteristics of individual cells. This makes possible the characterization and identification of different cell types in heterogeneous populations and the determination of purity of isolated subpopulations. In more advanced applications such as cell sorting, flow cytometers can also integrate the analysis results to a fluidic diversion system that enables sorting and collection of cells based on desired parameters.
The accuracy of FC analysis greatly depends on the use of suitable antibodies and optimized staining methods. Antibody titration and reactivity against the target antigen should be carefully determined and appropriate isotype controls should always be included in experimental designs. Rockland provides scientists with antibodies and supporting reagents tested and qualified for FC analysis including primary antibodies that recognize intracellular or cell surface cellular targets, secondary antibodies and isotype control proteins.
Buffers for Flow Cytometry
Primary Antibodies Recognizing Intracellular Targets
Staining of intracellular targets—often activated (i.e. phosphorylated) enzymes in key cell signaling pathways—for analysis by FC depend on the ability to successfully fix and permeabilize the cell membrane so that labeled antibodies can enter the cell (permeabilize) and interact with their target antigen.
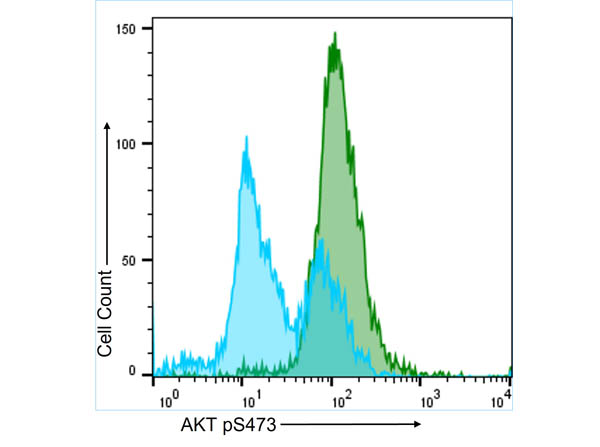
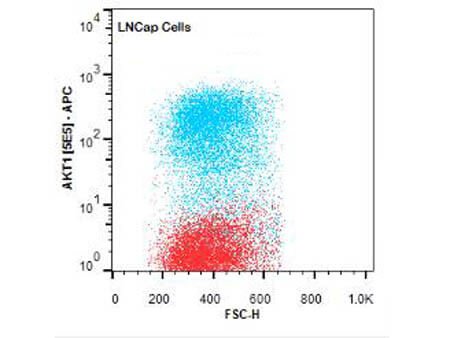
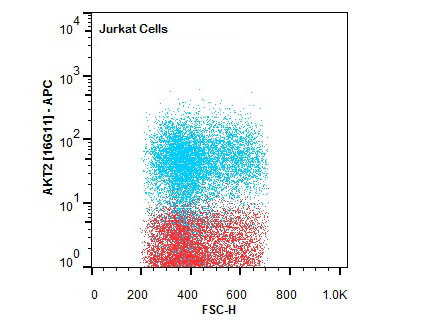
FC analysis of intracellular targets in permeabilized cells enable the study of key signaling pathways like AKT and NF-κB using Rockland’s primary antibodies conjugated to fluorescein (FITC), Phycoerythrin (PE), Allophycocyanin (APC), or biotin. Moreover, since highly specific antibodies for several PTMs such as phosphorylation and acetylation are also available as fluorescent conjugates, they allow FC analysis of relevant targets for epigenetics, stem cells, neuroscience, and cancer. Alternatively, when using non-conjugated primary antibodies, indirect fluorescent staining is also possible using a fluorescent secondary antibody.
Intracellular Flow Cytometry Antibodies
Primary Antibodies Recognizing Cell Surface Proteins
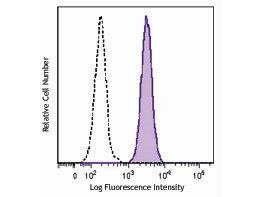
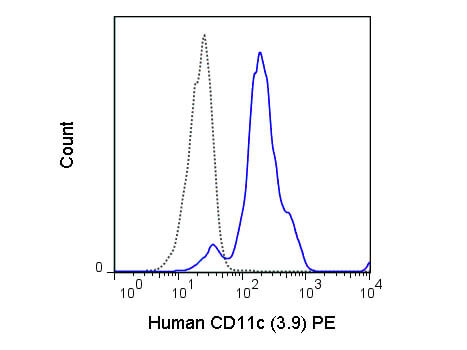
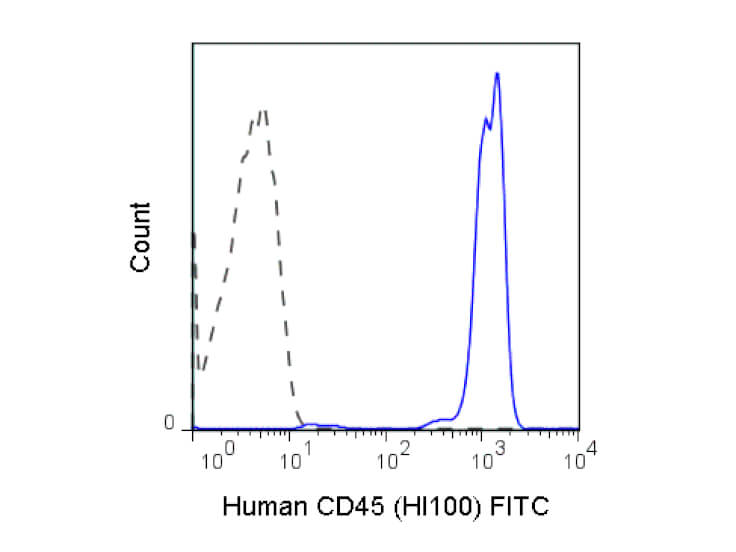
Conjugation of fluorescent molecules to antibodies constitutes a valuable tool for the detection of cell surface markers and other extracellular antigens by FC. A common approach involves the multicolor staining of individual antigens to analyze the expression of proteins on the cell surface only, or in combination with the analysis of intracellular targets. Rockland offers antibodies specific for human and murine cell surface proteins such as receptors for growth factors and cytokines, transporters, channels proteins and CD (cluster of differentiation) markers validated for immunophenotyping.
Immunophenotyping, the assessment of cell surface antigens using antibodies, is a widely used approach for characterization of cell populations. One of the most powerful techniques available for immunophenotyping is FC since it allows a simultaneous quantitative analysis of several cell surface antigens in either homogeneous or heterogeneous cell populations.
Flow Cytometry CD Antibodies
Secondary Antibodies Conjugated to FITC, PE or APC
Cell staining for FC with a non-labeled antibody that recognizes the target antigen requires treatment with a fluorophore-labeled secondary antibody, which exhibits specificity for the first antibody. Alternatively, the primary antibody can be conjugated to biotin and following binding to the target antigen, it is recognized using streptavidin conjugated to a fluorophore.
The use of secondary antibodies for FC has several advantages including amplification of the fluorescent signal and the possibility of using an unpurified and unlabeled primary antibody. Moreover, it also provides the opportunity to use biotinylated antibodies that can be detected using streptavidin conjugated to any of several fluorophores. Indirect staining with secondary antibodies can be easily combined with direct staining protocols using primary fluorophore-labeled antibodies and thereby seamless incorporated into multicolor staining experiments maintaining both specificity and flexibility.
Secondary Antibodies for Flow Cytometry
![Mouse IgG [H&L] Antibody CY5 Conjugated Pre-adsorbed](https://rocklandwebimagescdn.azureedge.net/webimages/610-110-121-MOUSE-IgG-Antibody-CY5-Conjugated-Pre-adsorbed-1-WBM-4x3.jpg)
Isotype Controls
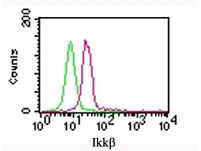
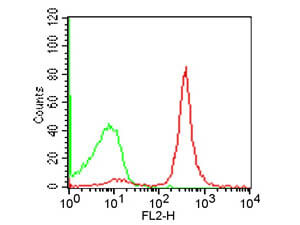
Robust data from FC greatly depends on the specificity of the antibody used. A reliable method for discrimination of background and authentic fluorescent signal is the use of a negative control antibody of the same isotype as the fluorophore-labeled primary or secondary antibody employed.
In order to obtain accurate quantitative data from FC experiments, it is critical to reliably confirm that the antibodies being used specifically recognize the intended target protein. This can be done by both optimizing the antibody dilution to be used and using the appropriate staining controls. Rockland’s isotype controls are reagents especially designed to help you determining staining background and non-specific antibody binding in FC assays. Isotype controls are available both as unconjugated or FITC-, PE- and APC-conjugated antibodies from different species including mouse, human and rat.
Isotype Controls for Flow Cytometry