Recombinant Anti-HER2 Fab Antibody
400-001-GY3
50 µg
Liquid (sterile filtered)
WB, ELISA, FC
Human
E. coli
Shipping info:
$65.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Product Details
Humanized Recombinant Anti-Human HER2 Fab fragment Antibody - 400-001-GY3
CD340, cluster of differentiation 340, ERBB2 (human), human epidermal growth factor receptor 2, HER2/neu, recombinant Monoclonal HER2, RabMAb HER2, Trastuzumab, Herceptin, Humanized Antibody, Recombinant Anti-HER2 Fab antibody, HER-2, Receptor tyrosine-protein kinase erbB-2, Metastatic lymph node gene 19 protein, Proto-oncogene Neu, p185erbB2
Recombinant Monoclonal
IgG Fab
E. coli
Target Details
ERBB2 - View All ERBB2 Products
Human
Recombinant Protein
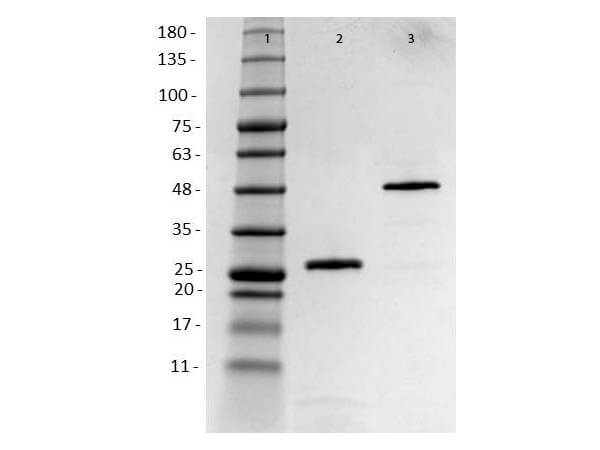
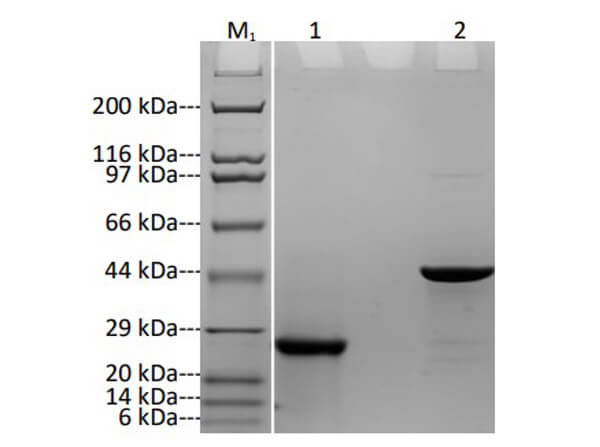
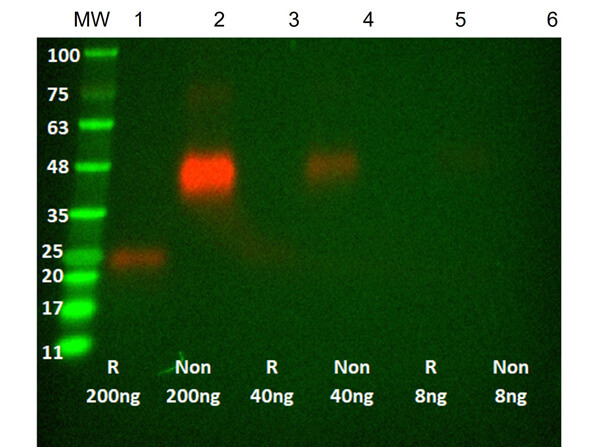
Humanized Recombinant HER2 Fab fragment was expressed in E. coli cells. The purity was estimated to be >90% by SDS-PAGE analysis.
Application Details
ELISA, FC, WB
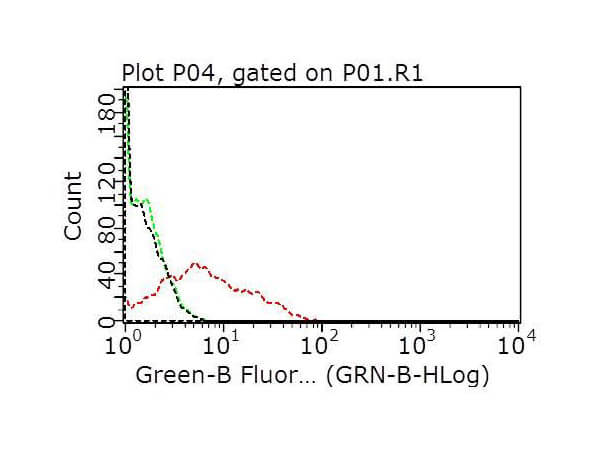
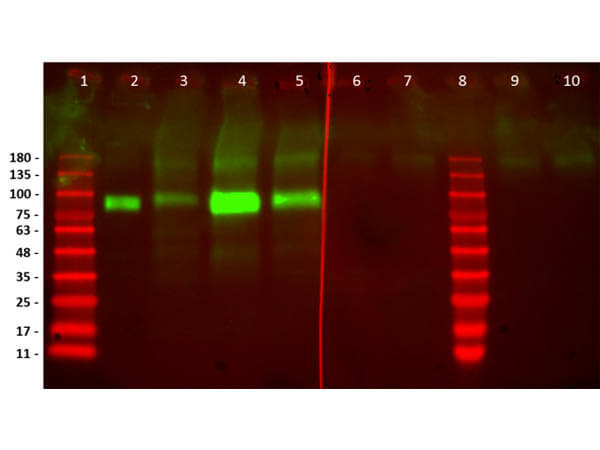
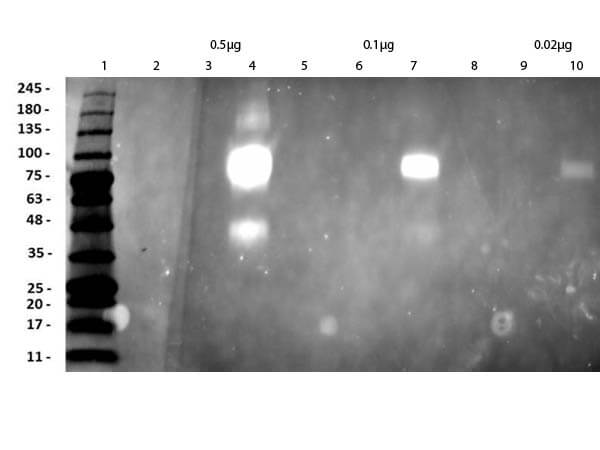
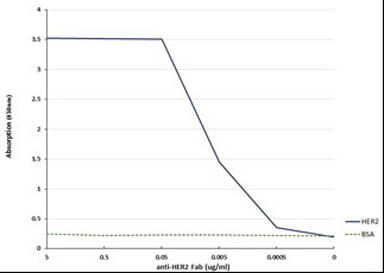
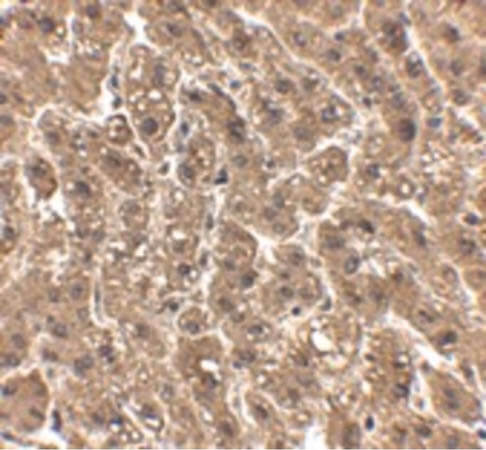
Humanized Recombinant Anti-HER2 Fab fragment Antibody has been tested for use in Flow Cytometry, Western Blot, and ELISA. This antibody recognizes structured HER2 and will work in western blot when the protein has not been denatured with DTT or bMe. Although not tested, this antibody could be useful in in IHC and in in-vivo and other cellular assays. Specific conditions for reactivity should be optimized by the end user.
Formulation
0.59 mg/ml by UV absorbance at 280 nm
0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2
0.01% (w/v) Sodium Azide
30% Glycerol
Shipping & Handling
Wet Ice
Store vial at -20° C. Dilute only prior to immediate use.
Expiration date is six (6) months from date of receipt.
HER2 (human epidermal growth factor receptor 2) is a member of the epidermal growth factor receptor (HER/EGFR/ERBB) family. It is also called ERBB2, CD340 or proto-oncogene Neu. The protein is a receptor tyrosine kinase located on the plasma membrane of cells. Activation of the tyrosine kinase promotes cell proliferation and suppresses apoptosis. HER2 can dimerize with any other member of the ErbB family, which results in auto phosphorylation of tyrosine residues and activation of signal pathways. HER2 is over expressed in a significant proportion of cancer patients, resulting in an increased disease recurrence and poor prognosis. Therefore an early diagnosis of the HER2 status of the tumors is necessary to decide the appropriate treatment. Besides in breast cancer HER2 is over expressed in variety of different tumors. The over expression of HER2 makes it a target for immunotherapy of certain tumor patients. The antibody Trastuzumab (Herceptin) which recognizes HER2 was approved for the treatment of HER2 positive breast cancer patients in 1998. The original approval was for treatment of HER2 positive metastatic breast tumors. Later studies have shown that Herceptin is also beneficial for early stage HER2 positive tumor patients and it is approved as an adjuvant treatment. Additionally the antibody is now approved also for other HER2 positive tumors (metastatic gastric adenocarcinoma). This antibody is made from a humanized Fab fragment making it specific, efficient, and effective. Humanized Recombinant Anti-HER2 Fab fragment Antibody is useful for researchers interested in Cancer research.
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.