TMB ELISA Peroxidase Substrate
27 References
TMBE-100
TMBE-1000
TMBE-8000
TMBE-20L
100 mL
1 L
8 x 1 L
10 x 2 L
Liquid - clear to very light blue colored liquid
Liquid - clear to very light blue colored liquid
Liquid - clear to very light blue colored liquid
Liquid - clear to very light blue colored liquid
ELISA
Shipping info:
$65.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Product Details
TMB ELISA Peroxidase Substrate - TMBE-100
3,3’,5,5’-Tetramethylbenzidine, TMB ELISA substrate, TMBE Substrate, chromogenic ELISA substrate
Target Details
pH: 4.0 +/- 0.2
Stability @ 18° to 26°C: PASS
Stability @ 4°C: PASS
QC Raw Material: PASS
Absorbance check of final product: PASS
Performance Data per ELISA: PASS
Application Details
ELISA
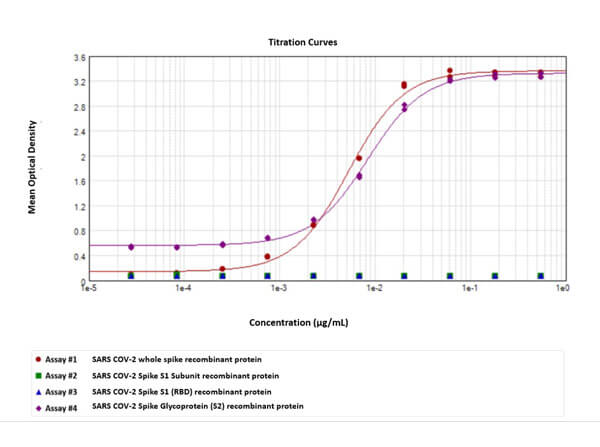
TMB ELISA Peroxidase Substrate comes ready to use. No dilutions are required. TMB ELISA Peroxidase Substrate will produce a soluble blue end product read at 370 nm or 655 nm. TMB ELISA Peroxidase Substrate incubation time will vary depending on the assay conditions.
Formulation
1x
Proprietary buffer with enhancer
Shipping & Handling
Ambient
Store container at 4° C prior to opening. Protect from moisture and light. No special shipping conditions or precautions are required.
Expiration date is one (1) year from date of receipt.
TMB (3,3´,5,5´-Tetramethylbenzidine) is a colorimetric substrate for peroxidase (HRP)-based enzyme immunoassays, notably ELISAs, and is known for producing a blue color that changes to yellow upon stopping the reaction with sulfuric acid. The color change is quantifiable at 450 nm. TMB´s high sensitivity makes it ideal for detecting low concentrations of analytes.
Fial I et al. (2025). Characterizing Antibodies Targeting Antisense Oligonucleotide Phosphorothioate and 2'- O-Methoxyethyl Modifications for Intracellular Trafficking and Biodistribution Studies. Nucleic Acid Ther.
Applications
EIA, ELISA
Brown GE et al. (2025). Liver portal fibroblasts induce the functions of primary human hepatocytes in vitro. Commun Biol.
Applications
EIA, ELISA
Escalera A et al. (2024). SARS-CoV-2 infection induces robust mucosal antibody responses in the upper respiratory tract. iScience.
Applications
EIA, ELISA
Rasquinha MT et al. (2024). Mt10 Vaccine Protects Diversity Outbred Mice from CVB3 Infection by Producing Virus-Specific Neutralizing Antibodies and Diverse Antibody Isotypes. Vaccines (Basel).
Applications
EIA, ELISA
Taira CL et al. (2024). Vaccination with O-linked Mannans Protects against Systemic Candidiasis through Innate Lymphocyte Populations. J Immunol.
Applications
EIA, ELISA
Liu JS et al. (2023). Decellularized Liver Nanofibers Enhance and Stabilize the Long-Term Functions of Primary Human Hepatocytes In Vitro. Adv Healthc Mater.
Applications
EIA, ELISA
Barker SJ et al. (2023). Targeting Transferrin Receptor to Transport Antisense Oligonucleotides
Across the Blood-Brain Barrier. Sci Transl Med.
Applications
EIA, ELISA
Pandit M et al. (2022). AMPK suppresses Th2 cell responses by repressing mTORC2. Exp Mol Med.
Applications
EIA, ELISA
Quach HQ et al. (2022). Detection of SARS-CoV-2 peptide-specific antibodies in Syrian hamster serum by ELISA. J Immunol Methods.
Applications
EIA, ELISA
Kukla, DA et al. (2022). Assessing the compatibility of primary human hepatocyte culture within porous silk sponges. Rsc Advances
Applications
EIA, ELISA
Kratzer B et al. (2021). Immunological imprint of COVID-19 on human peripheral blood leukocyte populations. Allergy
Applications
E, EIA
Pandit M et al. (2021). LKB1-PTEN axis controls Th1 and Th17 cell differentiation via regulating mTORC1. J Mol Med (Berl).
Applications
Undefined
Kukla DA. et. al. (2020). Microscale Collagen and Fibroblast Interactions Enhance Primary Human Hepatocyte Functions in Three-Dimensional Models. Gene Expr.
Applications
E, EIA
Kukla DA. et. al. (2020). Primary Human Hepatocytes Maintain Long-term Functions in Porous Silk Scaffolds Containing Extracellular Matrix Proteins. bioRxiv
Applications
E, EIA
Scharer CD et al. (2020). Antibody-secreting cell destiny emerges during the initial stages of B-cell activation. Nat Commun.
Applications
E, EIA
Shao W et al. (2020). Serum lipoprotein–derived fatty acids regulate hypoxia-inducible factor. J Biol Chem.
Applications
E, EIA
Harshbarger W et al. (2020). Convergent structural features of respiratory syncytial virus neutralizing antibodies and plasticity of the site V epitope on prefusion F. PLoS Pathog.
Applications
E, EIA
Ferdman J et al. (2019). Intra-seasonal antibody repertoire analysis of a subject immunized with an MF59®-adjuvanted pandemic 2009 H1N1 vaccine. Vaccine.
Applications
E, EIA
Haines, RR et al. (2018). The Histone Demethylase LSD1 Regulates B Cell Proliferation and Plasmablast Differentiation. Journal of Immunology (Baltimore, Md. : 1950)
Applications
EIA, ELISA
Tomita, K et al. (2018). Antibodies against adenovirus fiber and penton base proteins inhibit adenovirus vector-mediated transduction in the liver following systemic administration. Scientific Reports
Applications
EIA, ELISA
Barwick, BG et al. (2018). B cell activation and plasma cell differentiation are inhibited by de novo DNA methylation. Nature Communications
Applications
EIA, ELISA
Steff, AM et al. (2017). Pre-fusion RSV F strongly boosts pre-fusion specific neutralizing responses in cattle pre-exposed to bovine RSV. Nature Communications
Applications
EIA, ELISA
O'shannessy, DJ et al. (2016). Novel antibody probes for the characterization of endosialin/TEM-1. Oncotarget
Applications
EIA, ELISA
Parry, HM et al. (2016). Cytomegalovirus viral load within blood increases markedly in healthy people over the age of 70 years. Immunity & Ageing : I & A
Applications
EIA, ELISA
Ciferri et al. (2015). Antigenic Characterization of the HCMV gH/gL/gO and Pentamer Cell Entry Complexes Reveals Binding Sites for Potently Neutralizing Human Antibodies. PLOS Pathogens
Applications
E, EIA
Shimizu, K et al. (2014). Suppression of leaky expression of adenovirus genes by insertion of microRNA-targeted sequences in the replication-incompetent adenovirus vector genome. Molecular Therapy. Methods & Clinical Development
Applications
EIA, ELISA
O'shannessy, DJ et al. (2011). Characterization of the human folate receptor alpha via novel antibody-based probes. Oncotarget
Applications
EIA, ELISA
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.