Datasheet is currently unavailable. Try again or CONTACT US
Human IgM (myeloma) Fc5u
009-0131
1 mg
Liquid (sterile filtered)
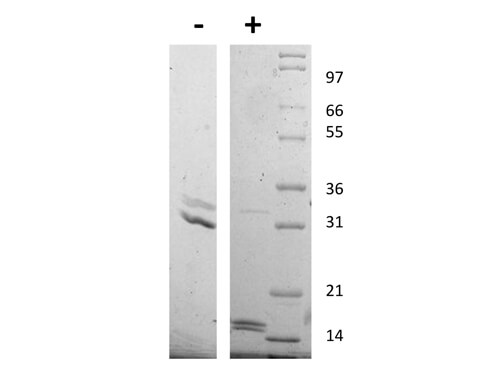
SDS-PAGE
Human
Shipping info:
$65.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Product Details
Human IgM (myeloma) Fc5µ Fragment - 009-0131
Human immunoglobulin M fragment; Human IgM Fc5µ
Human
IgM
Native Protein
Target Details
Human IgM (myeloma) Fc5µ fragment has been prepared from Human IgM myeloma protein by digestion with trypsin followed by column chromatography. Purity was assessed by SDS-PAGE and HPLC to be greater than 95%. A single precipitin arc was observed against anti-human IgM Fc5µ and anti-human serum when assayed by immuno-electrophoresis at a concentration of 20 mg/ml. No reaction was observed against anti-Trypsin, anti-human IgG F(ab´)2, anti-human IgG F(c), anti-human Kappa or anti-human Lambda.
Application Details
SDS-PAGE
Human IgM (myeloma) Fc5µ fragment has been tested in SDS-Page and can be used in Western Blotting and ELISA experiments as a control reagent.
Formulation
1.2 mg/mL by UV absorbance at 280 nm
0.1 M Tris Chloride, 0.5 M Sodium Chloride, pH 8.0
0.05% (w/v) Sodium Azide
Shipping & Handling
Wet Ice
Store vial at 4° C prior to opening. Human IgM (myeloma) Fc5µ fragment is stable 4° C as an undiluted liquid. Dilute only prior to immediate use. For extended storage mix with an equal volume of glycerol, aliquot contents and freeze at -20° C or below. Avoid cycles of freezing and thawing.
Expiration date is one (1) year from date of receipt.
Human IgM (myeloma) Fc5µ fragment consists of only the µ (mu) chain of the Fc fragment. Immunoglobulin M is the largest antibody isotype and the first to be secreted against an initial exposure to antigen. IgM is predominantly produced in the spleen. IgM is formed from covalently linking 5 immunoglobulins together. Due to this large size, IgM is typically isolated to the serum.
No test method can provide total assurance that the hepatitis B virus, hepatitis C virus, human immunodeficiency virus, or any other infectious agents are absent. Thus, all blood products, including purified proteins derived from human blood sources, should be handled at Biosafety Level 2 as recommended by the CDC/NIH manual entitled Biosafety in Microbiological and Biomedical Laboratories for potentially infectious human serum, blood specimens or proteins derived from same. Source material for the human blood product supplied to your facility has been tested for the detection of HIV antibody, Hepatitis B surface antigen, antibody to Hepatitis C, HIV 1 antigen(s), antibody to HTLV - I/II, and syphilis by FDA guidelines. All units were found to be non-reactive/negative for these tests. All human blood source material is collected in FDA licensed centers and is tested with FDA approved test kits.; This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.